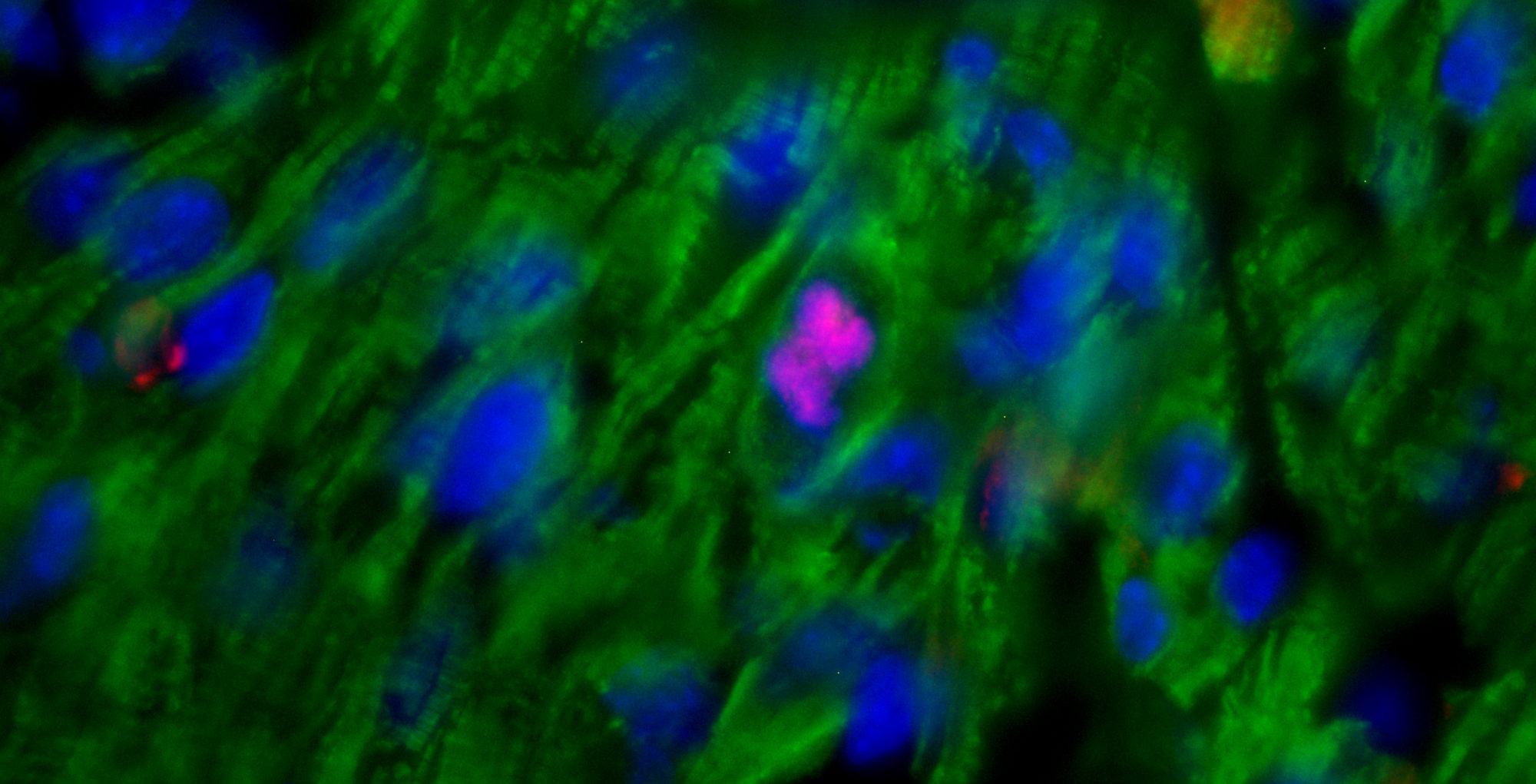
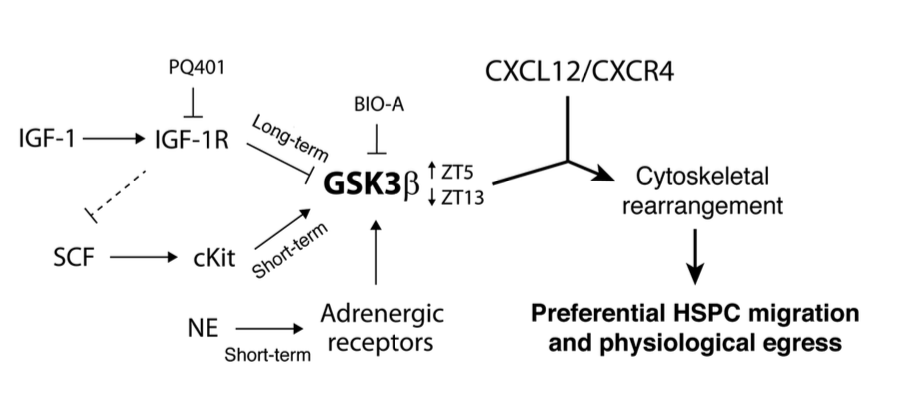
The role of corticosterone (Cort), the immune system’s major stress hormone, in the regulation of hematopoietic stem and progenitor cells (HSPCs) and their dynamic bone marrow (BM) microenvironment is currently unknown. We report that corticotropin-releasing factor receptor 1 (CRFR1) mutant mice with chronically low Cort levels showed aberrant HSPC regulation, having higher HSPC numbers and upregulation of the chemokine CXCL12, phenotypes that were restored by Cort supplementation. Expanded stromal progenitors known to support HSPCs were also observed in these low-Cort-containing mice. A similar phenotype was induced in wild-type (WT) mice by Metyrapone, a Cort synthesis inhibitor. Conversely, high Cort exposure induced HSPC apoptosis, reduced long-term BM repopulation and decreased stromal progenitor cell numbers. We documented circadian oscillations of Cort in WT BM but not in CRFR1 mutant mice, leading to diminished circadian BM CXCL12 fluctuations and increased number of circulating HSPCs in these mice. Finally, low Cort induced expansion of stromal progenitors, CXCL12 expression, HSPC proliferation and BM repopulation capacity, involving Notch1 signaling. This was associated with upregulation of the Notch ligand, Jagged1, in BM myeloid cells. Our results suggest that daily physiologic Cort oscillations are critical for balanced HSPC proliferation and function involving Notch1 signaling and their supportive BM microenvironment.
Vai all’articolo originale (in inglese): O Kollet*, Y Vagima*, G D’Uva, K Golan, J Canaani, T Itkin, S Gur-Cohen, A Kalinkovich, G Caglio, C Medaglia, A Ludin, K Lapid, E Shezen, A Neufeld-Cohen, D Varol, A Chen, T Lapidot. Physiologic corticosterone oscillations regulate murine hematopoietic stem/progenitor cell proliferation and CXCL12 expression by bone marrow stromal progenitors. Leukemia, 2013 (* equal contribution)
 Vincitori della poster session alla conferenza ISCS 2012 “The Young Investigators Stem Cell Meeting” con il progetto organizzato da Israel Stem Cell Society (2 Dicembre 2012. Tel Aviv, Israele).
Vincitori della poster session alla conferenza ISCS 2012 “The Young Investigators Stem Cell Meeting” con il progetto organizzato da Israel Stem Cell Society (2 Dicembre 2012. Tel Aviv, Israele).
 Abbiamo sviluppato Bitnos.com, un sistema operativo libero e basato sul web che permette di utilizzare le migliori applicazioni online, motori di ricerca e siti web gratuiti in campo biomedico. Con applicazioni e servizi on-line (noti anche come applicazioni web o webware), non è necessario scaricare e installare nulla. Tutti i servizi saranno direttamente disponibili per te in un solo click. Queste applicazioni e servizi funzionano indipendentemente dal sistema operativo che si sta utilizzando, in esecuzione tramite il browser come client. Hai solo bisogno di accedervi on-line.
Abbiamo sviluppato Bitnos.com, un sistema operativo libero e basato sul web che permette di utilizzare le migliori applicazioni online, motori di ricerca e siti web gratuiti in campo biomedico. Con applicazioni e servizi on-line (noti anche come applicazioni web o webware), non è necessario scaricare e installare nulla. Tutti i servizi saranno direttamente disponibili per te in un solo click. Queste applicazioni e servizi funzionano indipendentemente dal sistema operativo che si sta utilizzando, in esecuzione tramite il browser come client. Hai solo bisogno di accedervi on-line.